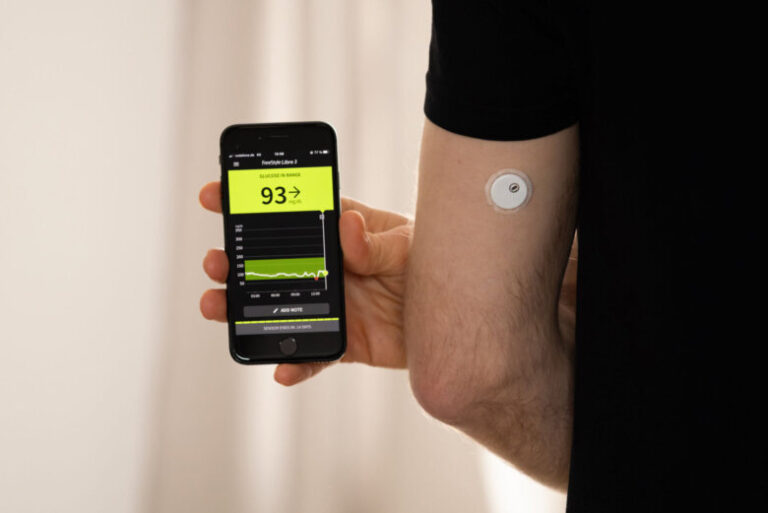
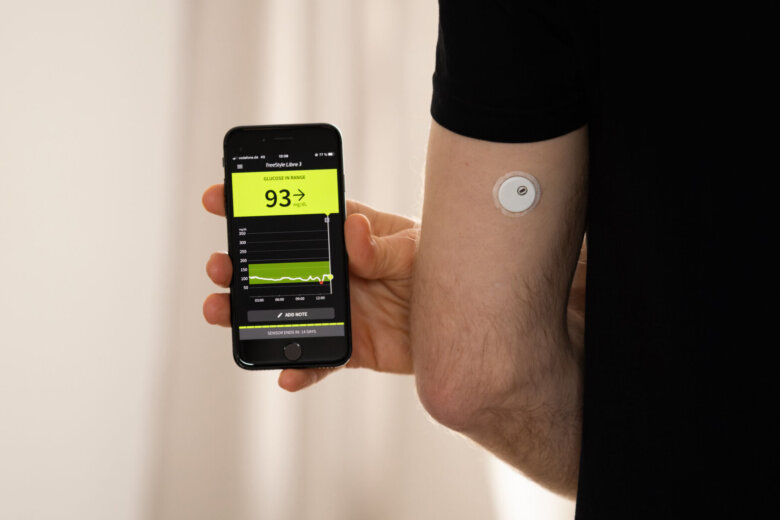
About 3 million FreeStyle Libre 3 and FreeStyle Libre 3 Plus sensors manufactured by Abbott were recalled in November, because they might give low sugar readings that are incorrect.
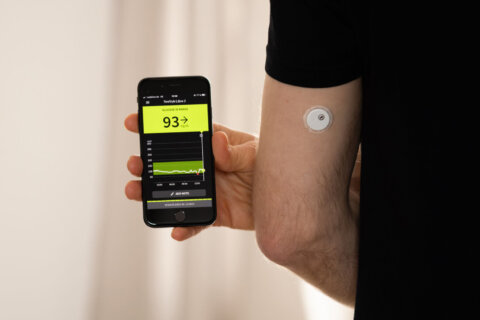
The number of reported problems tied to millions of continuous glucose monitoring sensors that were recalled last year has grown.
About 3 million FreeStyle Libre 3 and FreeStyle Libre 3 Plus sensors manufactured by Abbott were recalled in November, because they might give low sugar readings that are incorrect.
In addition to seven deaths associated with the problem, there have been 860 serious injuries reported as of Jan. 7. That’s up from 736 at the time of the recall.
Even when working as designed, devices like these have benefits and drawbacks, according to Dr. Vandhna Sharma, an endocrinologist at MedStar Montgomery Medical Center in Maryland.
Continuous glucose monitoring systems, or CGMs, are often worn on the arm or abdomen and send readings to an app on your smartphone.
Sharma called them “excellent tools for identifying glucose patterns over time.” They are, however, different from glucometers, also known as blood glucose monitors or BGMs. CGMs monitor glucose levels in the fluid that surrounds cells, while glucometers check the levels in your blood.
The reading you get from a CGM may have a lag time of as much as 15 minutes behind what you get from a glucometer.
“If the CGM gives you a reading that’s low, you have to measure with a finger-stick,” Sharma said. “With any medical device, you have to ensure accuracy, especially periods of hypoglycemia.”
Another thing to keep in mind is whether a reading you’re getting makes sense with what your body is telling you.
“Whenever I give a patient a continuous glucose monitor — a CGM — I always advise them to correlate … the CGM reading to how they feel,” she said.
Get breaking news and daily headlines delivered to your email inbox by signing up here.
© 2026 WTOP. All Rights Reserved. This website is not intended for users located within the European Economic Area.